Вирусные гепатиты остаются одной из важнейших социально значимых проблем, так как повышают риск развития цирроза печени и гепатоцеллюлярной карциномы [1–5]. Глобальная распространенность вируса гепатита С (ВГС) в педиатрической популяции сильно варьирует в разных странах, может достигать 0,13% и увеличивается по всему миру [6, 7]. В последние 2 десятилетия растет внимание к этиотропной терапии хронического гепатита C (ХГС) [8, 9]. Успех комбинированного лечения препаратами интерферона (ИФН) с рибавирином (Р) в отношении пациентов с клинически выраженным циррозом печени составляет 21,5% для всех больных и 11,3% для пациентов с ВГС генотипа 1, что говорит о низкой эффективности лечения для данной когорты [10, 11]. Безопасная и эффективная противовирусная безинтерфероновая терапия препаратами прямого противовирусного действия (ПППД) пациентов с репликативным вариантом хронической HCV-инфекции находит в России все более широкое применение в реальной клинической практике [12]. В апреле 2019 г. Европейским агентством по лекарственным средствам было одобрено применение пангенотипической фиксированной дозы глекапревира-пибрентасвира (ГЛЕ/ПИБ) для применения у подростков (12–17 лет), инфицированных ВГС, на основе высокой частоты устойчивого вирусологического ответа через 12 нед. (100%) в исследовании Dora I [13–15]. Среди применяемых в РФ ПППД для лечения детей преимущественной схемой выбора является ГЛЕ/ПИБ (ее применяют у 71% пациентов) [16]. Количество противопоказаний к использованию ПППД, по сравнению с комбинированной терапией препаратами ИФН и Р, значительно сузилось, появилась возможность применять этиотропную терапию с противопоказаниями к комбинированным схемам ранее трудных для лечения больных [17]. Профиль безопасности ГЛЕ/ПИБ соответствует профилю безопасности как у взрослых [18, 19], так и у детей [15. 20]. Очевидно, что современная противовирусная терапия ХГС при использовании ПППД-режимов – новый и надежный инструмент оптимального управления заболеванием и его эпидемиологией, а также важнейший компонент общих мероприятий, направленных на решение амбициозной и при этом вполне осуществимой задачи – полной элиминации ХГС на территории Российской Федерации [21].
Цель исследования – оценка эффективности и безопасности лечения ПППД мавирет у детей с ХГС в реальной клинической практике.
Материалы и методы
В исследование были включены 38 детей с ХГС в возрасте от 12 до 17 лет (средний возраст больных – 14,7 ± 1,9 года), из них 13 пациентов ранее получали противовирусную терапию (ПВТ) (ИФН-α-2b + ребетол или ПэгИФН-α-2b + ребетол), остальные 25 детей не получали ПВТ. До назначения ПППД пациенты наблюдались в детском инфекционном отделении городской клинической больницы им. Н.А. Семашко. Всем 38 больным был назначен мавирет. Длительность наблюдения за пациентами после окончания комбинированной терапии ИФН + Р – 7–14 лет (в среднем 6,4 ± 3,9 года) и непосредственно перед назначением мавирета – 3–15 лет (7,6 ± 4,1года). Препарат назначали в 2021 г. в ГБУЗ «Самарский областной клинический центр профилактики и борьбы со СПИД». Перед назначением терапии у родителей детей было получено информированное добровольное согласие на применение ПВТ ХГС. Всех больных обследовали, терапию проводили в соответствии с Методическими рекомендациями «Хронический вирусный гепатит С (ХВГС) у детей» (2021), «Стандартом специализированной медицинской помощи при хроническом вирусном гепатите C» (2012) в критериях диагностики и лечения и дозах для разных возрастов, поэтому по возрастному признаку дети не были разделены на группы. РНК ВГС определяли методом ПЦР (чувствительность – 10–15 МЕ/мл). Согласно Методическим рекомендациям, продолжительность лечения составила 8 нед. у 37 больных и 16 нед. у 1 ребенка (с ВГС генотипа 3, который ранее не ответил на комбинированную терапию). Эффективность терапии ПППД оценивали по результатам ПЦР на 12-й нед. после окончания ПВТ.
Статистический анализ проводили при помощи современных инструментов STATGRAPHICS Plus for Windows версии 5.1 и STATISTICA for Windows версии 10. Для оформления результатов исследований применяли пакеты из системы Microsoft Office 2021. Достоверность различий между данными в группах проверяли на основе расчета t-критерия Стьюдента.
Результаты
В группе больных ХГС, состоящей из 37 пациентов, была проведена терапия препаратом мавирет, который назначали детям с массой тела не менее 45 кг по 3 таблетки одновременно 1 раз в день с пищей (общая суточная доза: глекапревир 300 мг и пибрентасвир 120 мг) в течение 8 нед., 1 больному – в течение 16 нед. (с ВГС генотипа 3). ВГС генотипа 1в был выявлен у 21 (55,3%) пациента, 2а – у 1 (2,6%), 3а – у 16 (42,1%).
Были рассмотрены жалобы и клинические симптомы после проведения ИФН-терапии в катамнезе и до назначения ПППД (табл. 1). Они не отличались друг от друга, то есть пациенты были сопоставимы.
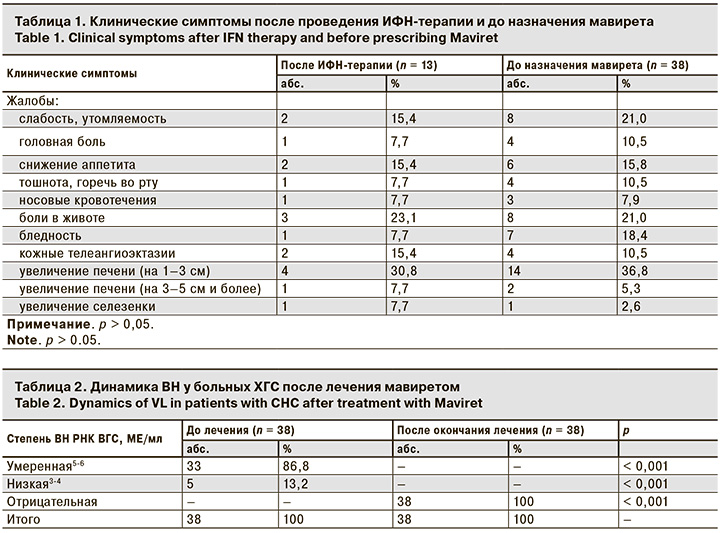
После окончания лечения ПППД клинические симптомы (слабость, снижение аппетита, боли в животе и др.) купировались.
Были изучены биохимические показатели больных ХГС после комбинированной терапии, а также до и после окончания лечения мавиретом (рис. 1).
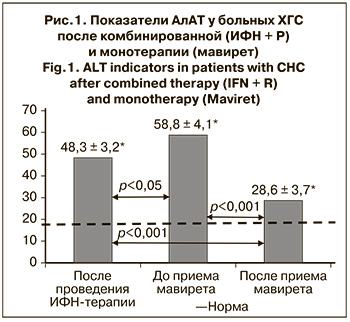
Повышенная активность АлАТ после проведения ИФН-терапии сохранилась у 2 (13,3%) больных ХГС. У 13 (34,2%) детей перед началом лечения мавиретом этот показатель был выше нормы (р < 0,05), а после окончания терапии ПППД у всех больных активность АлАТ нормализовалась – 28,6 ± 3,7 Ед/л (р < 0,001).
Динамика активности АсАТ была аналогична (рис. 2).
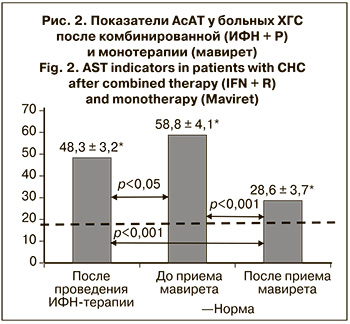
Снижения активности АсАТ до нормальных значений после проведения ИФН-терапии не произошло. С момента ее окончания до начала лечения мавиретом уровень АсАТ повысился с 45,8 ± 3,6 до 56,9 ± 4,9 Ед/л.
Повышенный уровень АсАТ после ИФН-терапии сохранялся у 3 (13,3) больных. Перед началом лечения мавиретом активность АсАТ повысилась у 11 (28,9%) больных (р > 0,05), а после его окончания достоверно снизилась до нормы – 23,3 ± 2,1 Ед/л (р < 0,001).
Была проанализирована вирусная нагрузка (ВН) до и после лечения мавиретом (табл. 2).
Продолжительность терапии составила 8 нед. у 37 больных и 16 нед. у 1 ребенка с ВГС генотипа 3. По окончании лечения у всех детей нормализировались биохимические показатели, через 12 нед. после окончания лечения у 100% пациентов наблюдался УВО.
Обсуждение
Целью ПВТ является достижение УВО, что подтверждается результатами лечения взрослых больных ХГС [22]. Аналогичные результаты как у детей, так и у взрослых представлены в работах других авторов. В исследованиях Л.Г. Горячевой и соавт. [15] с участием 67 детей подросткового возраста с ХГС, пролеченных препаратом ГЛЕ/ПИБ, новые схемы показали свою эффективность в 100% случаев. За время терапии у детей не зарегистрировано ни одного серьезного нежелательного явления, которое потребовало бы отмены лечения [23, 24], что подтверждается и нашими результатами. В исследование Т.В. Строковой [25] были включены 15 детей, которые принимали мавирет. УВО спустя 12 нед. после завершения лечения зафиксирован у всех пациентов. По данным В.В. Макашовой и соавт. [26], было проведено лечение с применением безинтерфероновых схем 70 взрослых больных ХГС, у которых наблюдалась высокая эффективность – до 100%, значимое повышение переносимости и безопасности терапии и минимальное влияние на качество жизни пациентов. В исследованиях И.Г. Никитина [21] отмечается, что при лечении взрослых больных ХГС препаратом ГЛЕ/ПИБ частота достижения УВО во всех группах составила 99,7%: среди пациентов с ВГС генотипа 3 – 98,4%, для всех остальных данный показатель достигал 100%.
Заключение
Результаты лечения детей с ХГС ПППД показали его высокую эффективность и безопасность. Сократилась продолжительность курса терапии. После окончания лечения у всех больных нормализировались биохимические показатели и отмечался УВО.
Расшифровка генома ВГС, создание новых ПППД, разрешение к применению и внедрение их в реальную клиническую практику позволят решить проблему ликвидации вирусных гепатитов в нашей стране, которую приняла Ассамблея ВОЗ в 2016 г. [27], а также исполнить Распоряжение Правительства РФ1 и Приказ Минздрава России2.



